|
The aluminum ion has a 3+ charge, while the fluoride ion formed by fluorine has a 1− charge.Rather than writing the formula as NaNaS, we shorten it by convention to Na 2S. Two sodium 1+ ions are needed to balance the 2− charge on the sulfide ion. To obtain a valence shell octet, sodium forms an ion with a 1+ charge, while the sulfur ion has a 2− charge.Ionic compound formed from iron and oxygen (assume the iron ion takes on a 3+ charge).Ionic compound formed from aluminum and fluorine.Ionic compound formed from sodium and sulfur. 
(You should be able to use a periodic table and the octet rule to determine likely charges for the main group elements.) Write the chemical formula for an ionic compound formed from the following elements. The formula Mg 2Cl 4 has balanced charges with the ions in a 1:2 ratio, but is not correct because it is not the lowest whole number ratio. 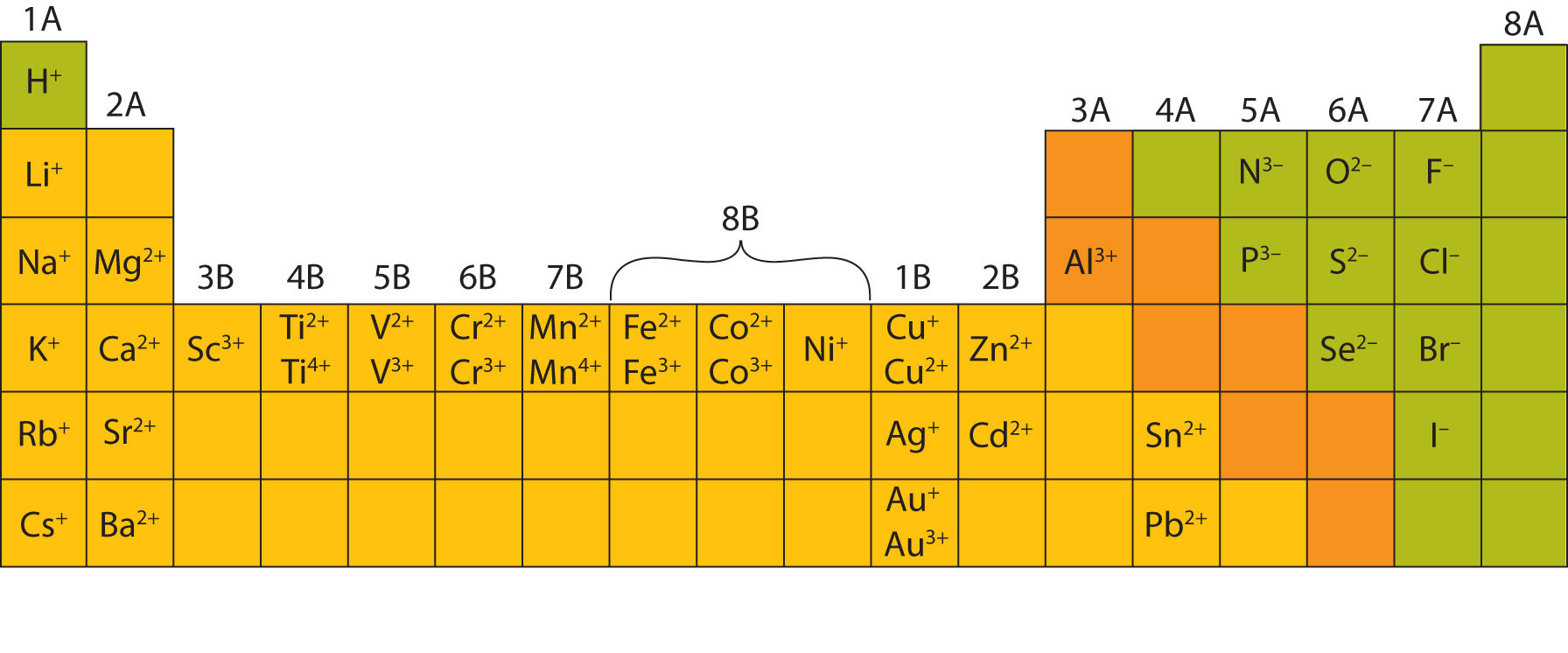
Rather, it exists as two individual chloride ions.) By convention, the lowest whole number ratio is used in the formulas of ionic compounds. Chlorine does not exist as a diatomic element in this compound. (Do not read the “Cl 2” part of the formula as a molecule of the diatomic elemental chlorine. This chemical formula says that there are one magnesium ion and two chloride ions in this formula. We could write the chemical formula for this ionic compound as MgClCl, but the convention is to use a numerical subscript when there is more than one ion of a given type-MgCl 2. Now the positive and negative charges are balanced. The easiest way to balance these charges is to assume the presence of two chloride ions for each magnesium ion: A magnesium ion has a 2+ charge, while a chlorine ion has a 1− charge:Ĭombining one ion of each does not completely balance the positive and negative charges. Now consider the ionic compound formed by magnesium and chlorine. Therefore, the proper formula for this ionic compound is MgO. Although both of these ions have higher charges than the ions in lithium bromide, they still balance each other in a one-to-one ratio. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Only one ion of each is needed to balance these charges. If we look at the ionic compound consisting of lithium ions and bromide ions, we see that the lithium ion has a 1+ charge and the bromide ion has a 1− charge. This rule is ultimately based on the fact that matter is, overall, electrically neutral.īy convention, assume that there is only one atom if a subscript is not present. To determine the proper formula of any combination of ions, determine how many of each ion is needed to balance the total positive and negative charges in the compound. Finally, the proper formula for an ionic compound always obeys the following rule: the total positive charge must equal the total negative charge. The ionic charges of all solutions were balanced by addition of sodium chloride, and. Remember that in an ionic compound, the component species are ions, not neutral atoms, even though the formula does not contain charges. of calcium and bicarbonate ions (Ca2+ HCO3. Second, charges are not written in a formula. Because most metals form cations and most nonmetals form anions, formulas typically list the metal first and then the nonmetal. First, the cation is written before the anion. The formula for an ionic compound follows several conventions. A crystal of sodium chloride, shown here, is a collection of alternating sodium and chlorine ions. Several examples are found in Table 3.3.1. The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as a single unit. 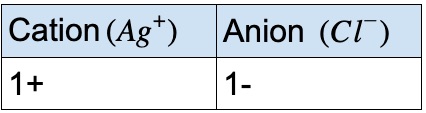
The formula of the carbonate ion is CO 32. The precise pattern depends on the compound. The carbonate ion (see figure below) consists of one carbon atom and three oxygen atoms and carries an overall charge of 2. A crystal contains a three-dimensional array of alternating positive and negative ions. Another role is in the chemistry of deflocculated mud, where bicarbonate ions prevent attachment of deflocculants such as lignosulfonate, onto clay edge charges.\) A Sodium Chloride Crystal.A second is the formation of calcium carbonate scale on surfaces by carbonate and calcium ion reactions.The ionic compound consists of a calcium cation along with two chloride anions reacting with each other to. One role is the corrosion of metals by acidic CO 2. The chemical formula of calcium carbonate is CaCl2.Carbonates play several important roles in water mud chemistry. If no component is lost from the system (such as CO 2 gas evolving), changing pH up and down continually reverses the relative proportion of the carbonate species. As pH rises above neutral, CO 3 –2 ions dominate. As pH rises from acidic toward neutral, HCO 3 – ions dominate. Carbonate chemistry involves a pH-dependent equilibrium between H 2O, H +, OH –, CO 2, HCO 3 – and CO 3 –2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
